Since the 1970s, biotinylation of antibodies for immunocytochemical applications has been used mainly to target antigens in cells and tissues. The general protocol to make an antibody-biotin conjugate with streptavidin was first described in 1994. It consisted of a biotinylated primary or secondary antibody applied to a sample of protein. The detection was carried out with labeled avidin or streptavidin. From then on, many authors have made modifications to the method but the basic concept remains attached to the importance of biotin-streptavidin reactions.
Biotin-Streptavidin reactions are highly selective and occur rapidly when both molecules are in solution. To make a biotin conjugate with streptavidin, attach biotin to one molecule, attach streptavidin to the second molecule, and then mix the solutions together.
What is Biotinylation?
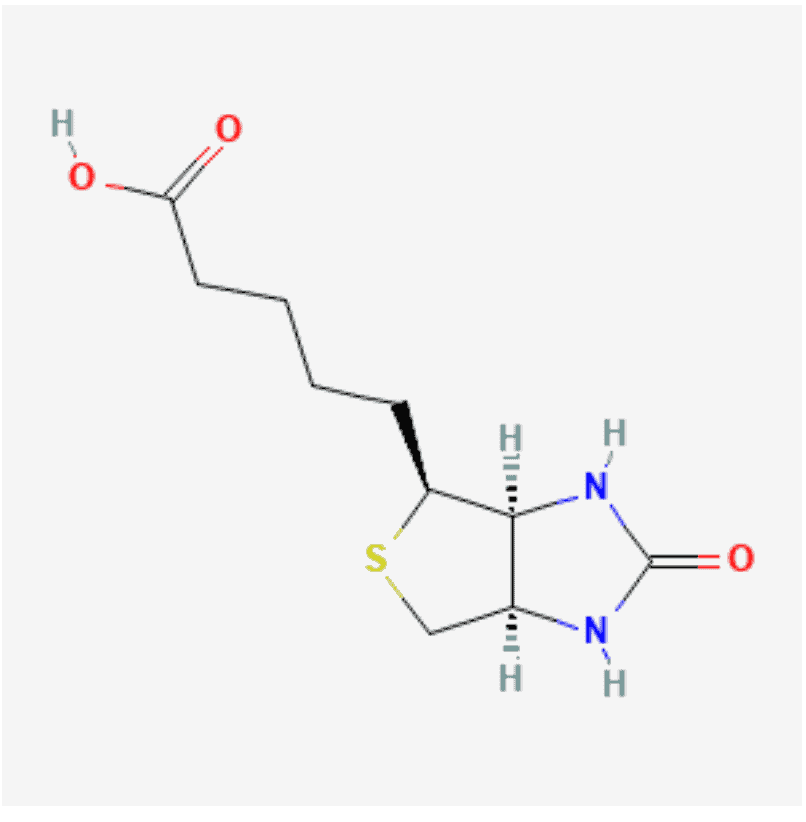
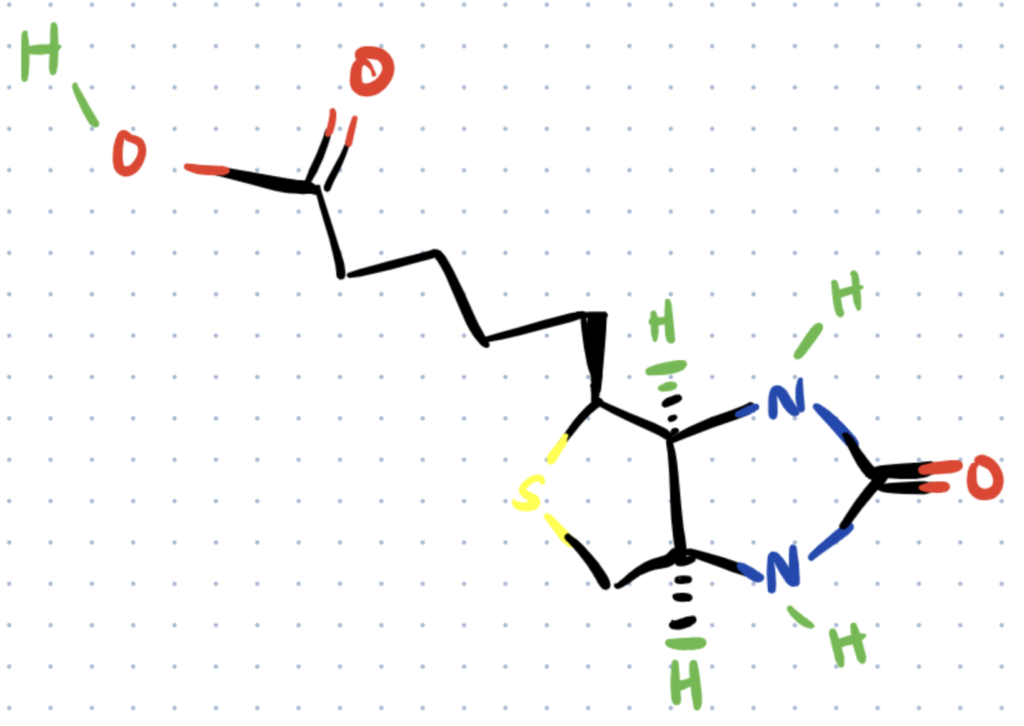
Biotinylation is the process of linking biotin, a water-soluble vitamin also known as vitamin H, with a molecule (typically a protein) either on an amino acid or in a carbohydrate fraction of the protein.
Avidin is a protein present in egg whites, which binds strongly to biotin. Streptavidin is a similar protein which originates in the bacteria Streptomyces avidinii which also binds strongly to biotin (source).
Biotin-Streptavidin Reactions
Biotin-Streptavidin reactions are highly selective and rapid because of their high affinity. Many authors have described this interaction as the strongest, non-covalent, biological interaction, with a dissociation constant or KD of 1.3×10-15 M (source).
Here, KD is the concentration of ligand (biotin) at which half of the ligand-binding sites on the protein (streptavidin) are occupied in the system equilibrium, and it is equivalent to the following equation:
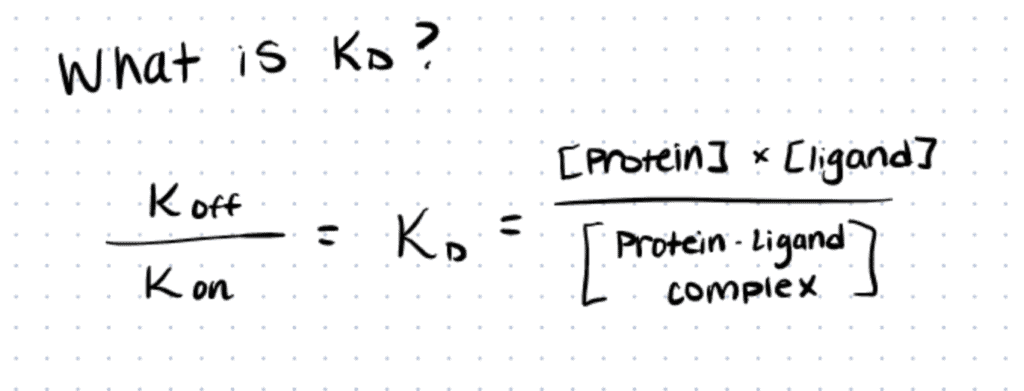
In this equation, P is the concentration of the protein, L is the ligand concentration, and P-L is the concentration of the protein-ligand complex once the equilibrium of the reaction is reached. As the units of the concentrations are represented in moles, it makes sense that the smaller the KD value, the greater the binding affinity of the ligand for its target (Source).
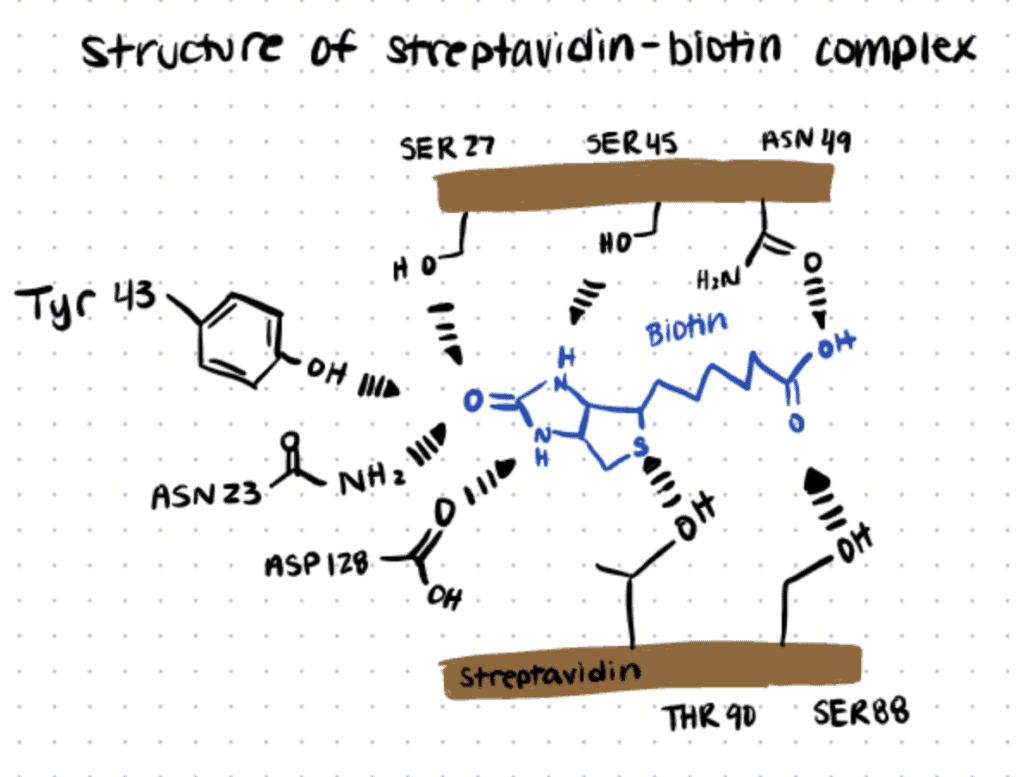
Streptavidin is a tetrameric protein, and each monomer bonds with one biotin molecule. The non-covalent bonds in the binding pocket are formed by a hydrogen-bonding network between biotin and Serine (SER), Asparagine (ASN), Tyrosine (TYR), and Threonine (THR) residues, as the following image shows.
How to Make a Biotin Conjugate with Streptavidin
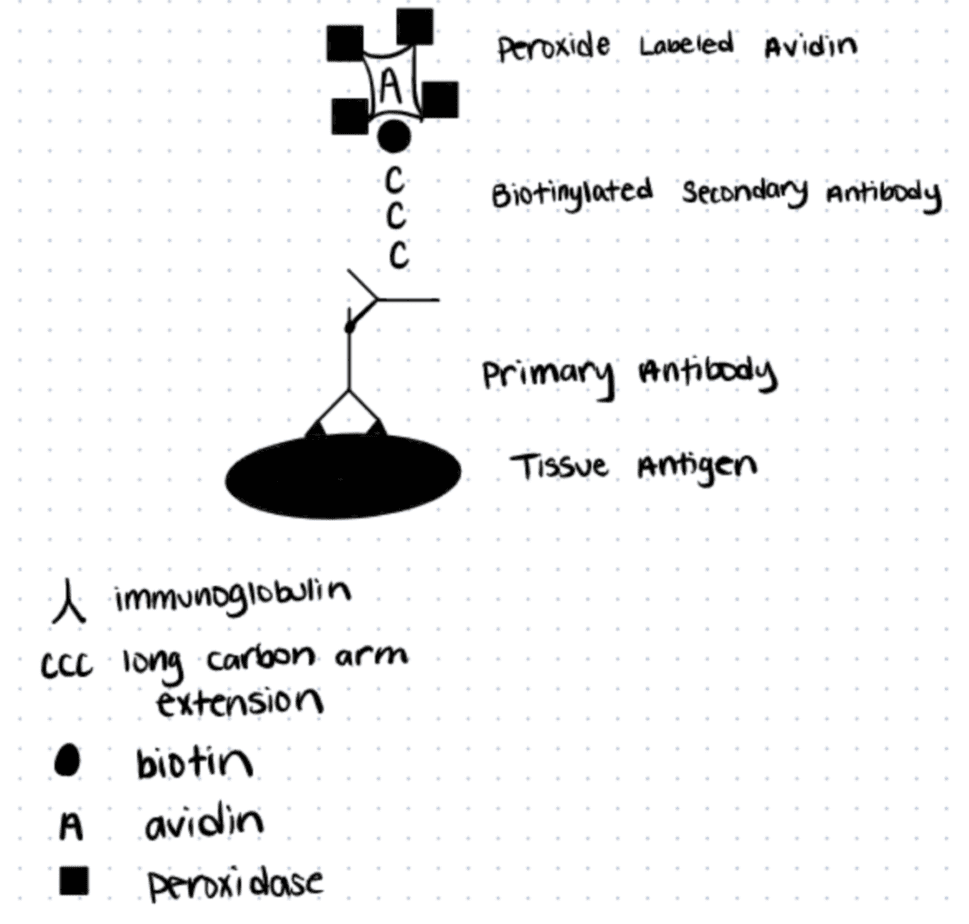
Labeled avidin binding (LAB), also called “the streptavidin binding method,” was first described by Gary Bratthauer as an alternative to the standard peroxidase-anti peroxidase (PAP) or avidin-biotin complex (ABC) methods. The main differences between those protocols and LAB included: a) using streptavidin instead of avidin because it works at neutral pH, and b) increased affinity towards biotin, as well as better sensitivity and resolution of the technique.
Below we discuss a general method for making a biotin conjugate with streptavidin. For more details read this article. You can also use alternative methods like Transglutaminase bioconjugation to biotinylate proteins.
Step 1. Block Slides with Serum
Incubate tissue slides overnight in 10% of serum in Phosphate-buffered saline (PBS). This step depends on the starting material, so the serum should be from the species from which the secondary antibody was generated.
Step 2. Add Primary Antibody
After removing the slides from the dish where the serum was incubated, add the primary antibody to the slide, covering it completely. Cover the humid staining chamber and incubate for at least 30 minutes. Rinse using PBS, repeating three times for a couple of minutes between subsequent washings. Read more
Step 3. Block Slide with Serum to Prevent Non-Specific Binding
Add more 10% serum in PBS, incubate for 10 minutes. Rinse with PBS and repeat the process and incubation one more time.
Step 4. Add Biotinylated IgG Secondary Antibody.
After absorbing the excess serum from each slide with absorbent paper towels, repeat step 2 with the secondary antibody, including at least a 30-minute incubation and subsequent rinsing with PBS.
Step 5. Add Labeled Streptavidin.
Add labeled streptavidin (ie. Streptavidin-peroxidase conjugate) at an appropriate dilution like 1:10000. Incubate for 30 minutes. Here we are creating the biotin conjugate with Streptavidin.
Step 6. Add Chromogen Solution.
Place the slides into a 3,3´diaminobenzidine tetrahydrochloride (DAB) chromogen solution, cover and incubate for 15 min, then wash with three changes of deionized water for 2 minutes each. Preparation and addition of DAB as a chromogen solution should be done with precaution as it is potentially carcinogenic. During incubation you might notice a brown precipitate which is created by the breakdown of DAB by peroxidase.
Step 7. Counterstain, develop nuclei blue, dehydrate and clear.
Counterstain with Mayer’s hematoxylin for 1-5 minutes. Rinse with deionized water, three changes, 2 minutes each. Develop the nuclei blue with a 10-second incubation in ammonium hydroxide in water. Rinse. Dehydrate with 100% ethanol, four changes for 2 minutes each. Clear the slides with xylene, four changes for 2 minutes each.
Step 8. Observe.
After coverslipping with Permount, see the tissue under a microscope.
Why Use An Antibody-Biotin Conjugate?
Use an antibody-biotin conjugate since it’s more specific, penetrates deeper into tissue, and is easier to purchase than antibody-protein conjugates like antibody-HRP and antibody-AP.
Biotin-Streptavidin conjugation is fast and efficient. In the above protocol, you’ll notice that you only need a secondary antibody that’s biotinylated. These are very easy to find compared to HRP or AP conjugates of secondary antibodies. They are also smaller than HRP or AP conjugates since biotin is a smaller molecule than HRP or AP proteins. This means that the secondary antibody can penetrate tissue better.
When compared to Western Blotting or ELISAs, using the antibody-biotin conjugate for tissue staining is better because it is challenging to find antibodies for doing western blots or ELISAs for all the tissue targets you can think of. ELISAs have the additional limitation of being cumbersome to carry out and have a high possibility of false-positive or false-negative. We go more in-depth about conjugated antibodies in our article, antibody conjugation methods. If you are looking for more information about western blots, consider our articles western blot exposure time and detection and how to choose western blot loading amounts.
Antibodies can be easily attached to other biomolecules like proteins, polymers, and carbohydrates using amines, carboxyls, and even thiols. Use these antibody conjugation kits to attach antibodies with other biomolecules.
Applications of Biotin Conjugation
Novel methods of biotin conjugation are continuously being developed for in vitro and in vivo applications. In recent years, there have been important developments in catalysis, cell biology, and proteomics, in addition to applications in the more established areas of detection, labeling and drug delivery. Below, you can read about three interesting applications.
Applications of biotin conjugation include the creation of biosensors, fluorescent labeling of live cells, and creation of quantum dot conjugates.
Antibody-biotin-Streptavidin-horseradish peroxidase (HRP) sensor
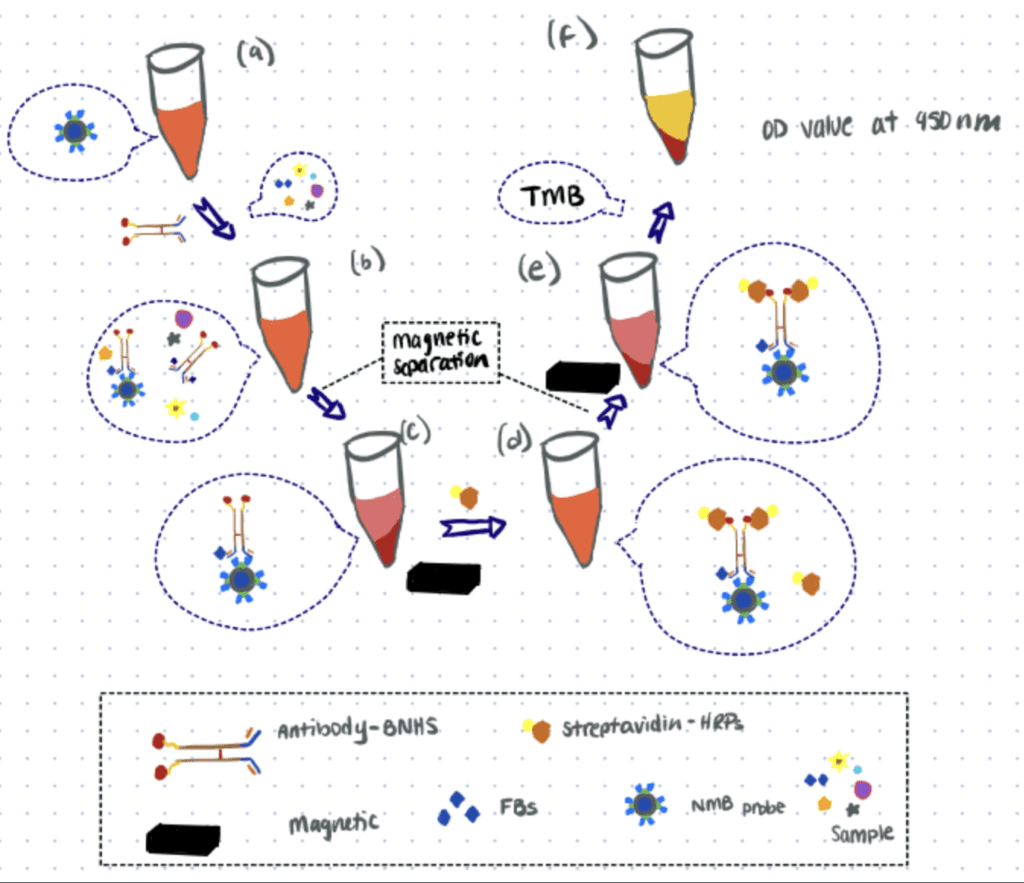
In this article, the researchers developed a sensor to detect fumonisins (FBs), carcinogenic mycotoxins that can affect many crops. This method is based on nanomagnetic beads (NMBs) and an antibody-biotin-streptavidin-HRP system.
For this application, a specific antibody for FBs is labeled with biotin-N-hydroxysuccinimide or biotin-NHS (BNHS) and streptavidin is then used as a bridge for loading HRP into the system. The NMB probe dispersed in solution competes with FBs in the sample for binding with antibody-BNHS. Then streptavidin-HRP is captured by antibody-BNHS. Finally, HRP oxidizes the substrate and generates visible signals that can be correlated to the concentration of FBs. Similarly, another application for biosensing is the use of hydrazine bioconjugation to detect breast cancer.
Fluorescent Labeling With Enzymatic Biotinylation
Fluorescent labeling of live cells is a method that enables the visualization of molecular events at the cell surface. To observe a fluorescent signal with any streptavidin-fluorophore conjugate, the cell surface must first be selectively biotinylated.
There are 3 common ways to biotinylate cells: Huisgen 1,3 cycloaddition, also known as click chemistry, Staudinger ligation, and enzymatic biotinylation by biotin protein ligase (BirA) produced by E.coli.
In this article, researchers described labeling cell surfaces by enzymatic biotinylation. Here, site-specific biotinylation was carried out on a 15-amino acid “acceptor peptide” (AP) by BirA. The researchers then used a streptavidin-Alexa 568 conjugate to observe the labeled proteins expressed on the surface of live HeLa cells. You can also use methionine selective bioconjugation for fluorescent labeling.
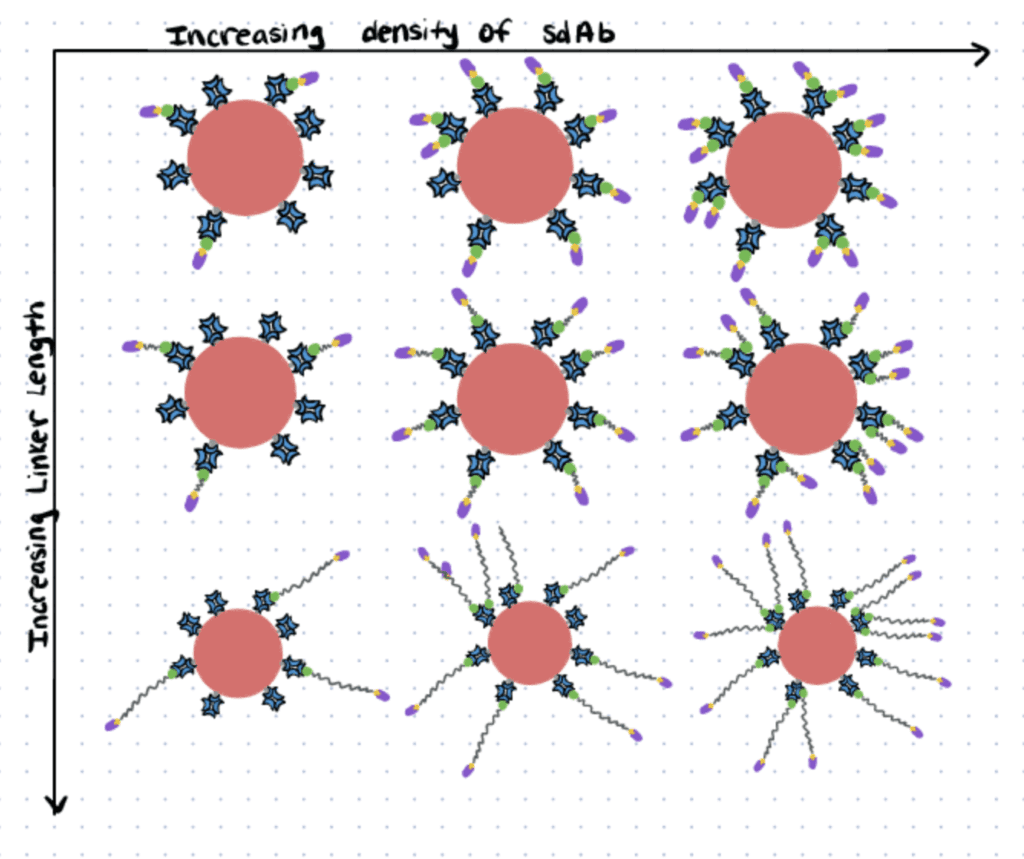
Dibenzocyclooctyne-poly(ethylene glycol)-biotin (DBCO-PEG-Biotin) linkers to couple Single-Domain antibodies (sdAbs).
In this article (sdAbs) were functionalized to target fluorescent nanocrystals (Qdots) to epidermal growth factor receptor (EGFR)-positive cells. The researchers focused mainly on engineering the nanoparticle surface to increase cell binding, with the ultimate aim of improving the delivery of therapeutics.
In order to couple the sdAbs to the Qdot surface, first, they coupled sdAbs with biotin through DBCO-PEG-biotin linkers, searching for the optimal PEG chain length.
Then they added these functionalized sdAbs to a Qdot-streptavidin conjugate, searching for the Qdot surface density -or sdAbs per Qdot- that enabled better cell binding.
Antibodies can be easily attached to other biomolecules like proteins, polymers, and carbohydrates using amines, carboxyls, and even thiols. Use these antibody conjugation kits to attach antibodies with other biomolecules.