The functional richness and structural vulnerability of peptides have often inspired researchers to implement bioconjugation strategies to create peptide-based drugs, polymer conjugates, and diagnostics. Currently, cysteine represents the most prominent target for direct peptide modification due to its inherent nucleophilicity compared to the side chains of other amino acids. We’ll discuss cysteine-based and other peptide bioconjugation protocols in this article.
Peptide-macromolecule bioconjugation methods include the use of bromopyridazinedione, pyridyl disulfide, and dithiodipyridine disulfide (DTDP).
Although cysteine residues are rarely encountered in native peptides, researchers can now engineer free, accessible cysteine to specific sites of interest through site-directed mutagenesis. A wide variety of reagents for cysteine-specific protein modification have been developed. Let’s get into the article and discuss several peptide bioconjugation methods.
A Method For Peptide Bioconjugation to Carbohydrates
Since Goebel and Avery (1929) emphasized the importance of conjugating sugars to a given protein, glycoproteins have been at the forefront of research in glycobiology. Despite being the most widely employed reagent for cysteine-selective modification of proteins, maleimide poses potential limitations (Schelté et al., 2000)
Most proteins and antibodies have readily available amines and carboxyls. You can label proteins and antibodies with these conjugation kits to save time and improve consistency between experiments.
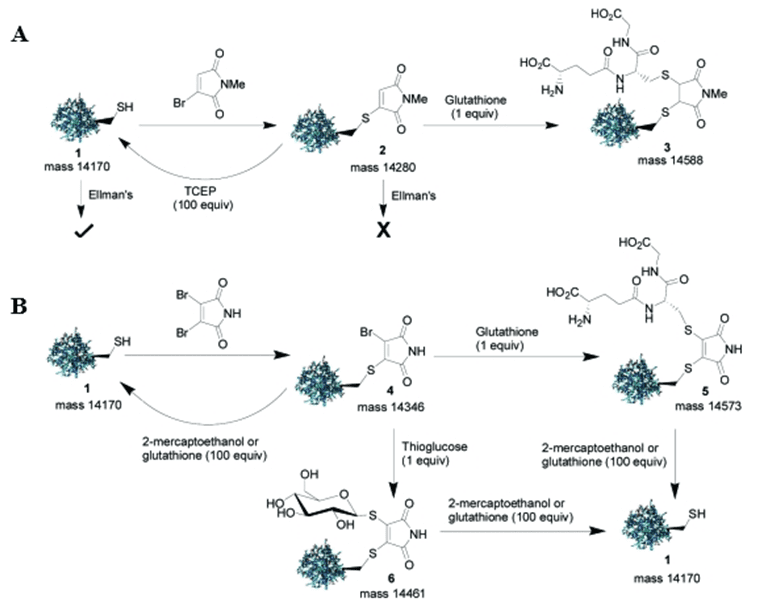
Recently, studies conducted by Tedaldi et al., 2009; Castañeda et al., 2013 have utilized a new class of functionalized maleimide-based reagent, bromomaleimide, for the selective and reversible modification of cysteine residues. With an established method in hand to generate both reversible and stable maleimide-bridged bioconjugates, the authors sought to demonstrate the applicability and versatility of this approach by using both the mono- and dibromomaleimide for reversible cysteine modification on proteins.
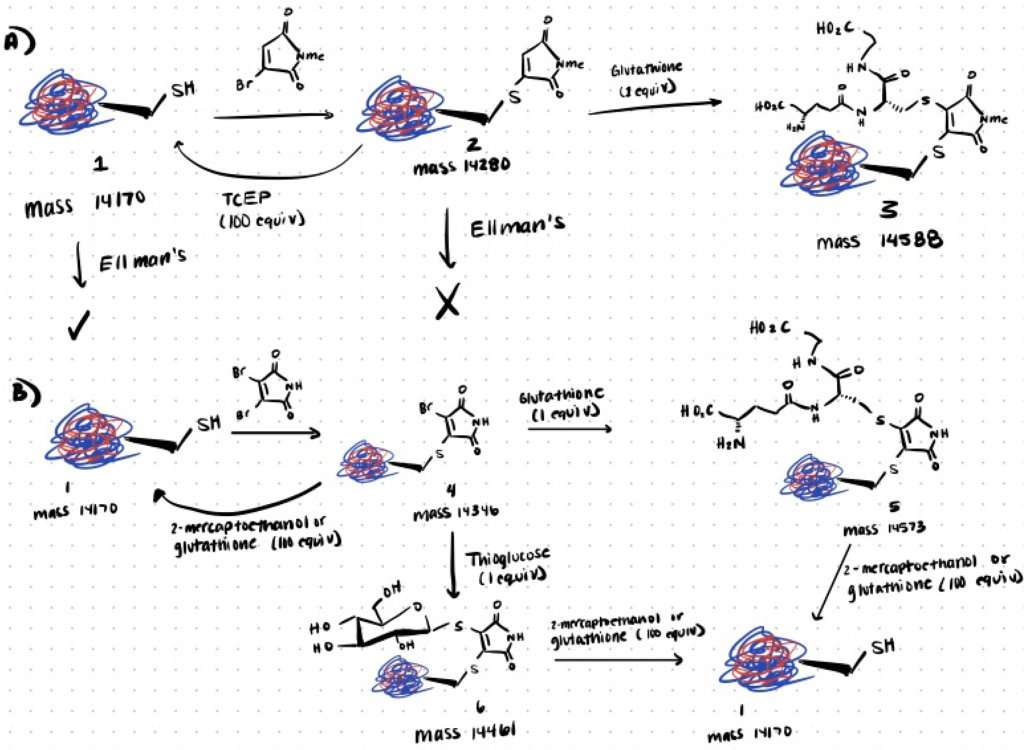
Step 1. Protein Modification With Monobromomaleimide
In this paper, the authors reacted the cysteine-containing Grb2-SH2 (L111C) protein mutant with the N-methylbromomaleimide at 0oC for 1 h. Then, following Ellman’s test, TCEP-mediated cleavage yielded a 85% conversion back to the initial protein. Thus demonstrating the potential of monobromomaleimides for the reversible covalent modification of cysteine residues in proteins. Further treatment with an excess of glutathione and 2-mercaptoethanol as the cleaving reagents also demonstrated that the protein-maleimide conjugate could undergo a second thiol conjugate addition to yield a 95% and 90% conversion rate, respectively. You can also read about thiol-mediated bioconjugation as it uses thiol-maleimide techniques to develop bioconjugates.
Step 2. Protein Modification With Dibromomaleimide
Similar to monobromomaleimides, no observed reaction with Ellman’s reagent occurred between the dibromomaleimide-protein conjugates since the functionalization occurred specifically at the cysteine residue of the protein. However, in this instance, the authors found that only the 2-mercaptoethanol- or glutathione-mediated cleavage reaction afforded clean conversion back to the free, unmodified protein.
Furthermore, the authors also proposed the use of dibromomaleimide as an alternative reagent for the selective modification of disulfides via the formation of a two-carbon maleimide bridge. The reaction was illustrated on the peptide hormone somatostatin. You can read more about it in the paper.
Peptide Bioconjugation to Lipids
Liposomal technology has significantly altered the field of modern drug delivery systems nowadays. However, challenges in clinical translation remain. One promising approach involves the use of surface functionalization to develop a class of ‘smart liposomes’ to fulfill the functional requirements.
Whereas most previous works deal with some thiol-reactive polymers and pre-formed nano-objects, a team of researchers designed a pH-dependent synthesis pathway using a pyridyl disulfide reagent to generate liposome-peptide bioconjugates with enhanced cellular targeting/uptake and endosomal bypass properties. In the paper, the authors synthesized two different lipopeptides based on the tbFGF and GALA-Cys peptides for cell targeting and endosomal escape, respectively.
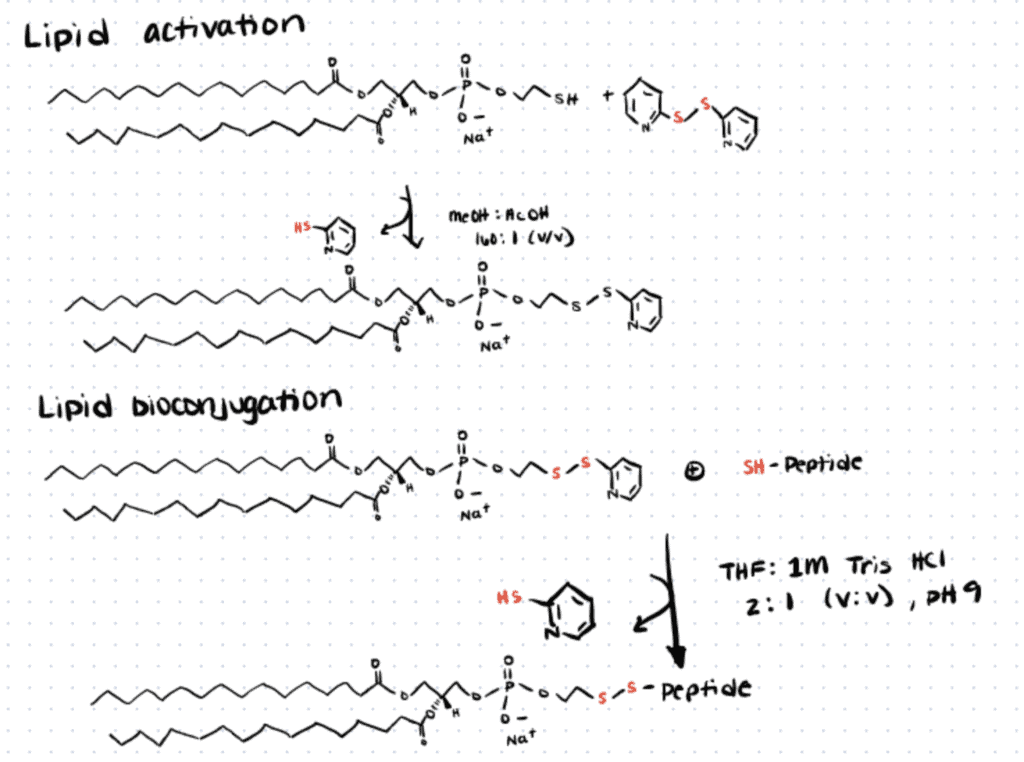
Step 1. Lipid Activation
To avoid non-specific unwanted reaction products, the authors first performed an irreversible reaction of the thiol-reactive 2-mercaptopyridine reagent with the thiol group of 1,2-dipalmitoyl-sn-glycero-3-phospho-thio-ethanol, DPTE (pH 4-5) to yield a reactive dissymmetric disulfide DPTE (aDPTE).
Step 2. Lipid-peptide Bioconjugation
Following purification of aDPTE, the authors then incubated the activated phospholipid with tbFGF and GALA-Cys at pH 9 to activate the cysteine residues of the protein. The reaction caused the disulfide bridge formed between DPTE and 2-mercaptopyridine to be substituted with the free cysteine groups of the proteins—producing a labile disulfide bond between the DPTE-peptide conjugates. You can read more about cysteine bioconjugation which goes over several techniques and applications in the conjugation of cysteine residues.
Step 3. Preparation And Characterization Of Liposomes
The liposomes were prepared according to the standard thin-film hydration method in chloroform buffer to form the multilamellar structure. The preliminary data obtained from the cell viability, confocal laser scanning microscopy, and luciferase assays of the liposomes decorated with both the DPTE-tbFGF and DPTE-GALA conjugates ensured the enhanced cellular uptake and cytosolic release of encapsulated cargo. Want to learn more about cellular uptake and bioconjugation? Read our related article.
Peptide Bioconjugation to DNA
During the last decade, single-molecule force spectroscopy (SMFS) has proven to be a powerful tool that can provide valuable insight into the previously inaccessible mechanical stability, ligand-binding, and protein folding properties of individual proteins. Although accurate and stable site-specific DNA-protein bioconjugation is a requirement for many SMFS applications, only a subset of the established methods is stable enough to find application in SMFS (Synakewicz et al., 2019; van der Sleen and Tych, 2021; Yang et al., 2020)
In this paper, Cecconi and his team (2008) pioneered a novel, site-specific DNA-protein bioconjugation method that extends the use of optical tweezers to study the unfolding/refolding mechanisms of individual globular proteins. By following the steps outlined below, the authors successfully integrated dsDNA handles on cysteine residues exposed on proteins to minimize non-specific interactions between the surfaces of the tethering beads.
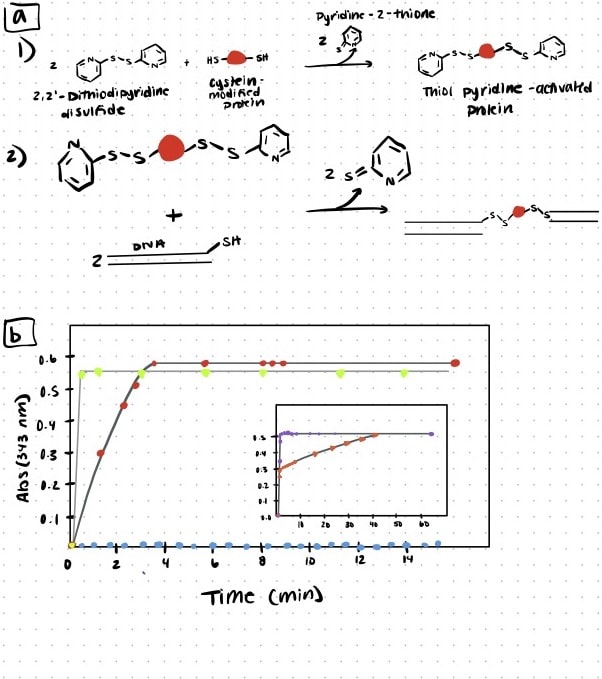
Step 1. DNA-protein bioconjugation
In this study, the authors attached two different types of dsDNA handles to the protein via the formation of a disulfide bond between a thiol group present at the end of the DNA molecule and a thiol group of a cysteine residue in the protein. To speed up the oxidation reaction, the thiol groups of the cysteine-containing proteins were first activated with DTDP (2,2’-Dithiodipyridine disulfide). The bioconjugation reaction was monitored spectrophotometrically at 343 nm via the release of the leaving group pyridine-2-thione
Step 2. Attachment Of Peptide-DNA Bioconjugates To Polystyrene Beads
In this research, the authors used two handle lengths, short (20/40 bp) and long (558 bp). Functionalization of the beads and introduction of compatible DNA handles was conducted according to streptavidin:biotin/digoxigenin:anti-digoxigenin chemistry. Because the attachment step occurs after the covalent modification of the target protein, the dsDNA handles can be tailored to a specific length.
Step 3. Characterization of bioconjugates
The resulting CD spectra and enzymatic activity studies revealed that the addition of DNA handles did not have a significant effect on the thermal stability, folding behavior, and structure of the tethered protein. The force-extension curves generated from optical tweezers measurements on monomeric and polymeric protein-DNA bioconjugates were successfully conducted in the physiologically relevant low-force regime—thereby allowing for the real-time monitoring of the mechanical behaviors and fluctuations between different molecular structures at lower tensions.
Peptide-Peptide Bioconjugation and Peptide-Protein Bioconjugation
We have only presented a mere fraction of the wide variety of peptide bioconjugation chemistry available today. Similar techniques as the ones we have already discussed can be utilized to create peptide-peptide and peptide-protein bioconjugates. However, it is more common for researchers to engineer peptides or proteins and then express them rather than to attach them together post-translation. This is because chemical methods for protein and peptide bioconjugation have lower yield than protein engineering methods.
Applications of Peptide Conjugation
Applications of peptide conjugation include the creation of peptide-based nanostructures, therapeutic peptides, and peptide biosensors.
Application 1. Peptide-Based Nanostructures
One of the most notable developments in the field of materials science includes the design of supramolecular materials consisting of self-assembling peptides. Originally described in 1993, this category of peptides can replicate what nature does—by assembling spontaneously into well-defined and functional structures according to their surrounding biochemical environment. The idea of exploiting known principles of peptide self-assembly has generated a wide variety of nanostructures with applications for specific biomedical uses, as reviewed by Lee et al., 2019 and Janković et al., 2021. Consider reading about electrochemical bioconjugation protocols, as it goes over the fabrication of nanostructures.
Application 2. Therapeutic Peptides
For many of us growing up in the 20th century, small-molecule drugs have historically been the pillars of our healthcare system. However, recently, the pharmaceutical industry has shown great focus on offering novel biologics to advance healthcare solutions and improve the patient experience. Although the physicochemical properties of peptides make them challenging to administer exogenously, various advances in the field of bioengineering and bioconjugation chemistry are paving the way for the increased use of peptides as biologics, particularly for the treatments of cancer, neurological disorders, and infectious diseases. With, over 80 different peptide drugs and vaccines that have attained FDA approval—and hundreds more currently in clinical trials—there is no doubt that the true magnitude of peptide therapeutics are expected to be much higher in the future.
Antibodies can be easily attached to other biomolecules like proteins, polymers, and carbohydrates using amines, carboxyls, and even thiols. Use these antibody conjugation kits to attach antibodies with other biomolecules.
Application 3. Peptide-based Biosensors
Peptide-based biosensors have a prominent role in the area of biosensor technology, particularly due to their inherent characteristics, diverse structures, and their potential for miniaturization when used in practice (Wee et al., 2005). Current bioconjugation strategies have provided means to fabricate novel peptide-based biosensors with various signal markers and biorecognition elements that can respond rapidly to environmental changes and stimuli. From odor sensors to detection of diseases—peptide biosensors play prominent roles in various commercial and clinical applications (Karimzadeh et al., 2018; Liu et al., 2015; Puiu and Bala 2018).
You can read more about the applications of biosensors in our articles bioconjugation to surfaces or polymer brush bioconjugation.
